- RECOMBIVAX HB
- Drug & Vaccine Interactions
- Active & Inactive Ingredients
- Clinical Trials, Efficacy Data & Duration of Protection
RECOMBIVAX HB#
RECOMBIVAX HB is a recombinant hepatitis B vaccine (a vaccine made using genetic engineering techniques) routinely given as a preventative against infection caused by the hepatitis B virus. It contains hepatitis B surface antigen or HBsAg (a protein found on the outer surface of the hepatitis B virus that triggers your immune system to respond) produced in yeast cells (Saccharomyces cerevisiae) using recombinant DNA technology (a method where scientists insert specific genes into an organism to make it produce desired proteins). A portion of the hepatitis B virus gene coding for HBsAg is cloned into yeast, creating a genetically modified organism that produces the surface antigen protein.
This was the first recombinant vaccine ever approved for human use and marked a paradigm shift from plasma-derived vaccines (older vaccines made from the blood plasma of infected individuals). The vaccine is adsorbed onto (attached to the surface of) aluminum hydroxyphosphate sulfate adjuvant (an aluminum-based substance added to vaccines to antagonize the immune system) and assumed to stimulate antibody production (the creation of protective proteins that help your body fight off the hepatitis B virus). However, the FDA insert states that the duration of protective effect in healthy vaccinees is unknown at present and the need for booster doses is not yet defined, despite decades of widespread use.
Mechanism of Action#
The aluminum adjuvant acts as an antagonist (an irritant that provokes a reaction), prolonging immune activation, and by triggering innate immune activation through inflammatory pathways. Protective antibody levels are defined as anti-HBs concentrations ≥10 mIU/mL. The vaccine is intended to establish immunological memory so that upon future exposure to hepatitis B virus, the immune system can rapidly mount an anamnestic (memory) response to prevent infection. However, vaccine-induced antibody titers decline over time, and while immune memory is presumed to persist, the actual duration of protection remains uncertain and unstudied as noted in the FDA insert. Therefore, without studies defining and proving protection duration, efficacy should not be claimed or stated.
What the Mechanism Affects#
The vaccine bypasses natural mucosal immunity barriers through intramuscular injection, forcing an unnatural and inferior route of immune activation. Residual formaldehyde from the manufacturing process (<15 mcg/mL) remains in the vaccine despite claims of safety based on endogenous formaldehyde production—however, injected formaldehyde bypasses normal metabolic pathways and detoxification mechanisms. The recombinant yeast protein residue (<1% yeast protein) from Saccharomyces cerevisiae fermentation may trigger allergic responses in sensitive individuals.
Critically, the duration and level of protection is unknown—the package insert explicitly states “the duration of the protective effect of RECOMBIVAX HB in healthy vaccinees is unknown at present and the need for booster doses is not yet defined.” Studies show antibody titers decline over time, with some vaccinated individuals losing detectable antibodies within 10 years. While immunological memory is theorized to persist, this has not been well established, and “breakthrough infections” have been well documented in vaccinated populations. Natural immunity from recovering from hepatitis B infection provides lifelong protection, whereas vaccine-induced immunity wanes, creating potential dependency on boosters of unknown necessity and timing. The vaccine does not protect against other forms of hepatitis (A, C, D, E) and provides no therapeutic benefit to those already infected with hepatitis B. Therefore, the vaccination of infants is fully unnecessary and without any proven or provable benefit.
Brand Names & Formulations#
- Pediatric/Adolescent: 5 mcg/0.5 mL (preservative-free)
- Adult: 10 mcg/1 mL (preservative-free)
- Dialysis: 40 mcg/1 mL (preservative-free)
All formulations are single-dose vials or prefilled syringes without preservatives (unlike some multi-dose formulations of other vaccines).
Prescribed For#
- Universal Infant Immunization: Assumed prevention of hepatitis B infection in all infants beginning at birth.
- Children and Adolescents: Catch-up vaccination for previously unvaccinated persons from birth through 19 years of age.
- Adults 20-59 Years: Universal vaccination has been recommended since 2022 by medical agencies.
- High-Risk Adults: Healthcare workers, persons with multiple sexual partners, homosexuals, injection drug users, persons with chronic liver disease, persons with HIV, travelers to endemic areas, and household/sexual contacts of HBsAg-positive persons.
- Dialysis Patients: Predialysis and dialysis patients aged ≥18 years using the Dialysis Formulation (40 mcg dose) due to reduced immune response in this population.
Contraindications, Precautions & Black Box Warning Status#
Absolute Contraindications & Major Precautions
Adverse Effects Profile#
Common Adverse Reactions (Clinical Trials)#
- Injection Site Reactions (17% of injections in adults): Soreness, pain, tenderness, pruritus, erythema, ecchymosis, swelling, warmth, nodule formation.
- Systemic Reactions in Infants/Children (10.4% of injections): Irritability (most frequent), fever (≥101°F oral equivalent), diarrhea, fatigue/weakness, diminished appetite, rhinitis.
- Systemic Reactions in Adults (15% of injections): Fatigue/weakness, headache, fever (≥100°F), malaise, nausea, diarrhea, pharyngitis, upper respiratory infection.
- Less Common: Sweating, achiness, sensation of warmth, lightheadedness, chills, flushing, vomiting, abdominal pains/cramps, dyspepsia, vertigo/dizziness, paresthesia, pruritus, rash, angioedema, urticaria, arthralgia (including monoarticular), myalgia, back pain, neck pain, shoulder pain, neck stiffness, lymphadenopathy, insomnia/disturbed sleep, earache, dysuria, hypotension.
Serious Adverse Reactions (Post-Marketing)#
- Anaphylaxis & Severe Hypersensitivity: Anaphylactic/anaphylactoid reactions including shock, bronchospasm, urticaria reported within first few hours after vaccination. Life-threatening and requires immediate emergency intervention. Serum-sickness-like syndrome of delayed onset (days to weeks) including arthralgia/arthritis, fever, dermatologic reactions (urticaria, erythema multiforme, ecchymoses, erythema nodosum).
- Autoimmune Diseases: Systemic lupus erythematosus (SLE), lupus-like syndrome, vasculitis, polyarteritis nodosa reported post-marketing.
- Neurological Disorders: Guillain-Barré syndrome, multiple sclerosis, exacerbation of multiple sclerosis, myelitis including transverse myelitis, seizures, febrile seizures, peripheral neuropathy including Bell’s Palsy, radiculopathy, herpes zoster (reactivation), migraine, muscle weakness, hypesthesia, encephalitis, optic neuritis, tinnitus, visual disturbances, uveitis.
- Dermatological Reactions: Stevens-Johnson syndrome (severe and potentially life-threatening), alopecia (hair loss), petechiae, eczema.
- Hematological Abnormalities: Thrombocytopenia (low platelet count, can cause bleeding), increased erythrocyte sedimentation rate.
- Gastrointestinal/Hepatic: Elevation of liver enzymes, constipation. Paradoxically, a hepatitis B vaccine causing liver enzyme elevations raises questions about hepatic effects.
- Musculoskeletal: Arthritis (distinct from arthralgia), pain in extremity.
- Psychiatric: Irritability, agitation, somnolence (beyond normal infant reactions).
- Cardiovascular: Syncope (fainting), tachycardia (rapid heartbeat).
- Other Vaccines’ Reactions: Keratitis (corneal inflammation) has been reported with another recombinant hepatitis B vaccine but not specifically with RECOMBIVAX HB.
Drug & Vaccine Interactions#
Active & Inactive Ingredients#
RECOMBIVAX HB is a suspension of subunit viral vaccine derived from HBsAg produced in genetically modified yeast cells. The formulation varies by preparation:
Active Substance#
- Hepatitis B Surface Antigen (HBsAg), adw subtype: Produced from recombinant Saccharomyces cerevisiae yeast cells containing cloned hepatitis B virus gene. The HBsAg is harvested from fermentation cultures and purified through physical and chemical methods. Amount per dose varies by formulation:
- Pediatric/Adolescent: 5 mcg per 0.5 mL dose
- Adult: 10 mcg per 1 mL dose
- Dialysis: 40 mcg per 1 mL dose
Adjuvant (Immune Stimulator)#
- Amorphous Aluminum Hydroxyphosphate Sulfate: Approximately 0.5 mg aluminum per mL of vaccine (0.25 mg per 0.5 mL pediatric dose, 0.5 mg per 1 mL adult dose). This aluminum compound serves as an antagonist to the immune system, creating inflammation and is recognized as aneurotoxin. Research has documented aluminum adjuvant biopersistence in macrophages, translocation to lymph nodes and other organs including brain tissue, induction of inflammatory responses, and associations with autoimmune conditions (macrophagic myofasciitis, ASIA syndrome), chronic fatigue syndrome, and potential neurodevelopmental effects. Aluminum interferes with calcium metabolism, increases oxidative stress, binds to ATP, and competes with iron and magnesium. While regulatory agencies assert safety based on a comparison to dietary aluminum exposure, this comparison is problematic because injected aluminum bypasses normal gastrointestinal barriers and detoxification mechanisms. And because these claims of safety have never been proven scientifically.
Manufacturing Residuals#
- Formaldehyde: <15 mcg/mL residual concentration. Used during manufacturing to inactivate and detoxify bacterial components and stabilize the product. While formaldehyde is produced endogenously in the body as part of normal metabolism, injected formaldehyde bypasses normal metabolic pathways and detoxification mechanisms. It is known to damage DNA and be carcinogenic. Regulatory agencies assert safety based on comparison to endogenous formaldehyde levels, but this comparison is disputed as it ignores route of administration and lack of metabolic buffering and lacks scientific basis in study.
- Yeast Protein: <1% residual yeast protein from Saccharomyces cerevisiae fermentation and purification process. May cause allergic reactions in yeast-sensitive individuals. The vaccine is contraindicated in those with yeast allergy.
Inactive Ingredients (Excipients)#
- Sodium Chloride: Isotonicity agent (maintains osmotic balance with body fluids)
CRITICAL NOTE ON VACCINE PRODUCTION: RECOMBIVAX HB is produced using recombinant DNA technology, making it the first recombinant vaccine approved for human use. A portion of the hepatitis B virus gene coding for HBsAg is cloned into Saccharomyces cerevisiae yeast. The yeast is grown on complex fermentation medium consisting of yeast extract, soy peptone, dextrose, amino acids, and mineral salts. The HBsAg protein is released from yeast cells by cell disruption and purified through a series of physical and chemical methods. The purified protein is treated in phosphate buffer with formaldehyde and then coprecipitated with alum (potassium aluminum sulfate) to form bulk vaccine adjuvanted with amorphous aluminum hydroxyphosphate sulfate. This manufacturing process involves genetic modification of organisms and chemical treatments, leaving residues that remain in the final product despite purification efforts.
Clinical Trials, Efficacy Data & Duration of Protection#
This vaccine has not been evaluated in placebo-controlled clinical trials. In the absence of such trials, efficacy cannot be causatively established, as observed outcomes cannot be reliably distinguished from placebo effects, bias, confounding variables, or natural disease progression.
❗ Furthermore, without controlled comparative data, the safety profile of this product has not been comprehensively characterized. Rare, delayed, or context-specific adverse effects may remain undetected, and the true incidence of potential risks cannot be accurately determined.
📌 Accordingly, claims of therapeutic benefit or assurances of safety should be interpreted with caution until validated by rigorous, randomized, placebo-controlled studies designed to meet established scientific standards.
CRITICAL DISCLOSURE ON DURATION OF PROTECTION#
The package insert explicitly states: “The duration of the protective effect of RECOMBIVAX HB in healthy vaccinees is unknown at present and the need for booster doses is not yet defined.” This represents a fundamental uncertainty about the vaccine’s long-term efficacy despite nearly 40 years of widespread use since its approval in 1986. While the vaccine has been administered to billions of people worldwide, the actual duration of protection remains unstudied and unknown for most populations.
Efficacy in Neonates (Peripartum Exposure)#
The protective efficacy of three 5 mcg doses of RECOMBIVAX HB is based on a study that is extremely difficult to access—it exists only in a hard-to-find reference book rather than being available in standard online databases. This lack of accessibility alone raises concerns about transparency and independent verification. The FDA insert claims that among infants who received one dose of hepatitis B immune globulin (HBIG) at birth followed by the recommended three-dose regimen of RECOMBIVAX HB, chronic infection had not occurred in 96% of 130 infants after nine months of follow-up. However, a thorough examination reveals multiple methodological flaws and analytical problems that fundamentally undermine the validity of these efficacy claims.
Major Methodological Flaws#
1. Flawed Assumption About Infection StatusThe study makes a critical logical error by implicitly assuming that the absence of detectable hepatitis B surface antigen (HBsAg) is equivalent to the prevention of infection. This assumption conflates two distinct biological states: the absence of a detectable marker versus the true absence of viral infection. This represents a fundamental misunderstanding of what the data actually demonstrate.
The presence or absence of HBsAg is a surrogate marker, not a direct measure of infection status. Without more comprehensive virological testing, particularly HBV DNA detection, the study cannot distinguish between genuine infection prevention and several alternative scenarios, including viral suppression, occult infection, or temporary clearance followed by later reactivation.
2. Absence of Proper Study Controls#
The study lacks essential elements of rigorous clinical trial design. There was no randomization of participants, which means potential confounding factors were not controlled. There was no blinding of outcome assessment, creating risk of observer bias. Most critically, there was no placebo-control group or untreated comparison group, making it impossible to establish true vaccine efficacy or rule out that observed outcomes would have occurred naturally without intervention.Without these controls, we cannot definitively attribute the assumed reduction in chronic infection to the vaccine itself rather than to other factors such as natural immune clearance, the protective effect of HBIG alone, or characteristics of the study population that made them less susceptible to chronic infection.
3. Selection Bias and Attrition Problems#
The study suffers from significant attrition bias. Infants who were lost to follow-up before completing the study were excluded from the final analysis. This creates a survivorship bias where only infants who survived early life complications and whose families remained engaged with the study protocol were included in the efficacy calculations.This exclusion is particularly problematic because adverse events, complications, or families dissatisfied with the intervention may be disproportionately represented among those lost to follow-up. The analysis therefore systematically overrepresents compliant families with positive outcomes while underrepresenting potential negative outcomes. When long-term follow-up at four to five years was attempted, only very small subsets of the original cohort could be assessed (as noted in Table VIII of the source material), further compounding this bias.
4. Plausible Alternative Explanations Not Ruled Out#
Because of the lack of appropriate controls and comprehensive testing, several alternative explanations for the observed outcomes cannot be excluded:Passive Antibody Masking: The HBIG administered at birth provides passive immunity that may temporarily suppress HBsAg detection without actually preventing infection. The virus could remain present in the body at levels below detection thresholds or in areas not tested.
Natural Immune Clearance: A significant proportion of infants exposed to hepatitis B may naturally clear the infection without progression to chronic disease, particularly those exposed perinatally rather than in utero. Without an untreated control group, we cannot determine what proportion of the observed “prevention” would have occurred naturally.
Lower Maternal Infectivity: The study cohort may have consisted of mothers with lower viral loads or different HBV variants that are less efficiently transmitted. If the study inadvertently selected for less infectious mothers, this would inflate apparent vaccine efficacy.
5. Problematic Cord Blood Exclusion#
The researchers excluded cord blood HBsAg results from their analysis, reasoning that cord blood HBsAg status correlated poorly with later infection outcomes. While this may seem reasonable, it creates serious interpretive problems:Underestimation of In Utero Infections: Excluding cord blood data may lead to systematic underestimation of infections that occurred before birth, which vaccines administered after birth cannot prevent.
Circular Reasoning Risk: If early classification of infection status depends on later outcomes, there is a risk of circular reasoning where the researchers are essentially defining infection based on whether the vaccine appeared to work, rather than using independent criteria.
6. Reliance on Inadequate Surrogate Endpoints#
The study primarily relies on two surrogate markers: the absence of detectable HBsAg and the presence of anti-HBs antibody titers. Neither of these endpoints is sufficient to support claims of infection prevention:HBsAg Negativity Is Not Proof of No Infection: The absence of detectable HBsAg does not exclude occult hepatitis B infection, where viral DNA remains present in hepatocytes or other tissues despite negative serological markers. Studies using sensitive molecular techniques have documented occult HBV infection in individuals who test negative for HBsAg.
Anti-HBs Levels Do Not Equal Sterilizing Immunity: While antibody titers may correlate with some level of protection in population studies, individual antibody levels do not guarantee sterilizing immunity (complete prevention of infection). An individual can have robust anti-HBs titers and still experience breakthrough infection.
No HBV DNA Testing Performed: Most critically, the study did not perform HBV DNA testing. This is the standard they use for detecting active viral replication but fail to use it to prove vaccine efficacy.
7. Statistical Power Issues and Inappropriate Interpretation#
The study is underpowered for several key comparisons. Some critical analyses involve very small event counts: only 5 HBsAg carriers in the recombinant vaccine group compared to 16-22 in plasma-derived vaccine groups. With such low numbers of events:High Risk of Type II Error: The study lacks sufficient statistical power to detect real differences between groups, meaning that true differences in efficacy may exist but go undetected.
Misinterpretation of Non-Significance: The researchers frequently interpret statistically non-significant results as evidence of equivalence between vaccine types. This is a fundamental statistical error. The absence of evidence for a difference is not evidence of absence of difference. Properly establishing equivalence requires specific equivalence testing with predefined margins, which was not performed.
Confidence Intervals Likely Too Wide: With small sample sizes, confidence intervals around efficacy estimates would be very wide, indicating substantial uncertainty about the true effect size, yet this uncertainty is not adequately communicated.
What Effect Has the HepB Vaccine Had?#
Population-Level Impact Analysis#
The CDC frequently presents historical data on pediatric hepatitis B incidence to support universal infant vaccination programs. However, a critical examination of these data reveals substantial questions about the vaccine’s population-level effectiveness and the justification for universal newborn administration.
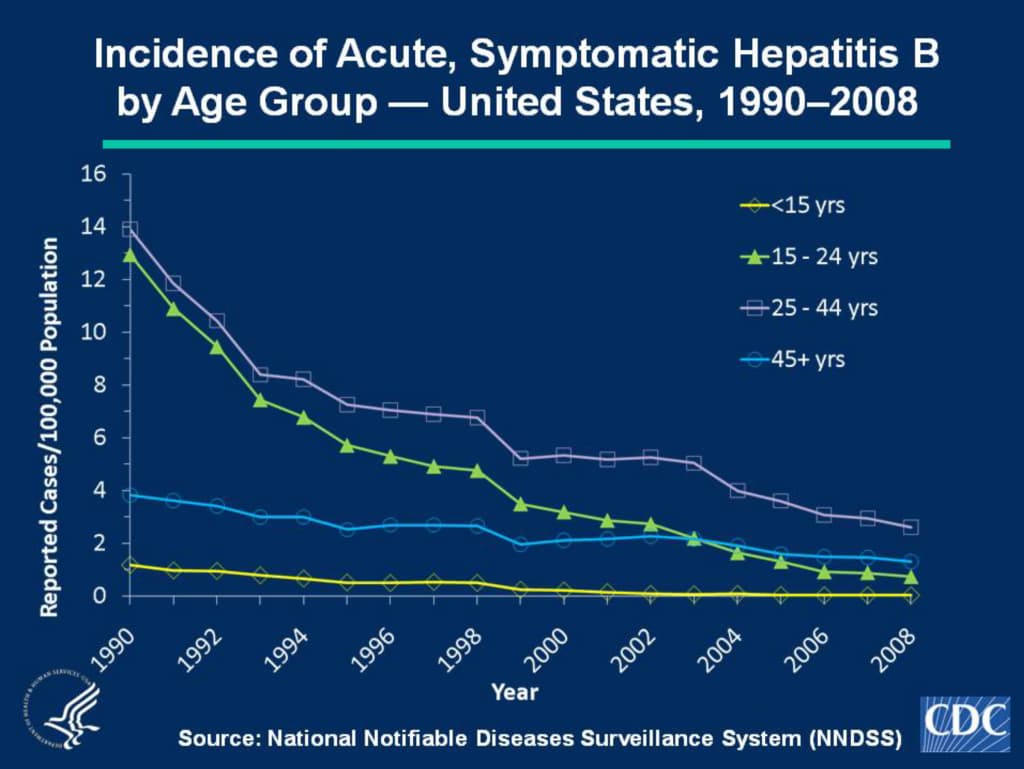
Historical incidence data frequently cited in vaccine promotion materials
Critical Analysis of Historical Trends#
According to CDC documentation, vaccine coverage rates for hepatitis B did not exceed 70% until after 1995. Even assuming, for the sake of analysis, that all decline in pediatric hepatitis B incidence after 1981 is attributable to vaccination (an assumption not supported by controlled evidence), the data present several concerning features:
- Age Group Aggregation: The CDC’s historical data aggregates all children up to 15 years of age, obscuring age-specific trends and conflating populations with vastly different risk profiles. This methodological approach systematically distorts the apparent impact on infants specifically.
- Number Needed to Treat: Even accepting the most favorable interpretation of the historical data, the number needed to treat (NNT) to prevent one case of acute hepatitis B in the birth cohort would be approximately 60,000 infants. This means 59,999 infants receive a medical intervention from which they derive no benefit, while being exposed to potential risks.
- Alternative Explanations: The decline in pediatric hepatitis B cases coincided with multiple public health interventions including blood supply screening, improved infection control practices, and changing patterns of intravenous drug use. The specific contribution of infant vaccination cannot be isolated from these confounding factors without controlled studies.
Recent Surveillance Data#
Beyond the methodological concerns with historical trend data, examination of recent surveillance data from the National Notifiable Diseases Surveillance System reveals that incidence rates in the pediatric population have not shown the sustained decline one would expect from an effective universal vaccination program implemented decades ago:
Incidence of Acute, Symptomatic Hepatitis B
by Age Group — 2016–2023#
The surveillance data from 2016-2023 reveal several troubling patterns that contradict expectations for an effective universal vaccination program implemented more than three decades ago:
- Persistent Cases Despite High Coverage: Acute symptomatic hepatitis B cases continue to occur in all pediatric age groups, including in infants under one year of age who should theoretically be the most protected given near-universal vaccination at birth during this period.
- No Clear Declining Trend: The data show year-to-year fluctuations rather than a consistent downward trend. Total pediatric cases have not declined below baseline levels observed when the vaccine was first introduced, and in some years have been significantly higher.
- Failure of Herd Immunity Theory: If the vaccine provided robust, long-lasting protection and achieved sufficient coverage, one would expect to see near-elimination of pediatric cases. The uptick of cases is very concerning.
- Statistical Noise vs. Public Health Impact: With such low absolute case numbers (1-5 total cases per year across all pediatric age groups in a population of millions), the data are dominated by statistical noise. Small fluctuations appear dramatic in percentage terms but represent minimal actual public health impact, calling into question whether universal infant vaccination can be justified for what amounts to fewer than a handful of cases annually.
FUNDAMENTAL RISK-BENEFIT QUESTION: The population-level data present a profound ethical dilemma. To potentially prevent one case of acute hepatitis B in the pediatric population requires vaccinating approximately 60,000 infants—all of whom are exposed to the intervention's potential risks with no personal benefit. This becomes particularly concerning when recent surveillance data show no clear evidence of declining incidence despite decades of universal vaccination, and when the vast majority of vaccinated infants face negligible hepatitis B exposure risk during the period of demonstrable vaccine protection. The continued occurrence of cases in vaccinated cohorts, combined with evidence of waning immunity and the absence of placebo-controlled efficacy data, raises fundamental questions about whether universal newborn vaccination represents sound public health policy or an intervention that exposes many to potential harm for minimal population-level benefit.
Please report any adverse events you experience during or after vaccination:- VAERS (Vaccine Adverse Event Reporting System): 1-800-822-7967 or www.vaers.hhs.gov - A passive surveillance system that captures only an estimated 1-10% of actual adverse events according to various studies. Both healthcare providers and patients/parents can report.
- Merck Sharp & Dohme Corp: 1-877-888-4231 - Direct manufacturer reporting
Duration of Protection - The Critical Unknown#
Despite nearly 40 years of use, the duration of protection remains officially "unknown":
- Antibody Decline: Multiple studies document that anti-HBs antibody titers decline over time. Some individuals lose detectable antibodies (<10 mIU/mL) within 10 years post-vaccination. One study showed 29.9-62.4% of vaccinated 15-year-olds had undetectable antibodies.
- Immunological Memory Theory: Regulatory agencies and vaccine manufacturers claim that even when antibodies become undetectable, "immunological memory" persists and will generate an anamnestic (rapid memory) response upon exposure to hepatitis B virus. However, this theory has limitations:
- Not all individuals mount anamnestic responses when challenged with booster doses years later
- Some studies show anamnestic response rates declining from >90% at 15 years to 73-88% at 30-35 years
- Absence of anamnestic response may indicate loss of immune memory
- No long-term studies beyond 35 years to assess protection in middle-aged and elderly adults
- Breakthrough Infections: Cases of hepatitis B infection have been documented in previously vaccinated individuals, particularly in areas with high endemic rates. One study found HBV infection rates increased with age in vaccinated cohorts, from 2.3% at age ≤16 years to 8.4% at age 25 years.
- Limited Long-Term Studies: The longest follow-up studies are from Alaska Native populations vaccinated in 1981 with plasma-derived vaccine (not RECOMBIVAX HB specifically), showing 35-year follow-up data. Even these studies show concerning decline in protective antibody levels and anamnestic responses.
CRITICAL ANALYSIS: The unknown duration of protection represents a fundamental flaw in the hepatitis B vaccination program. Millions of infants are vaccinated at birth based on assumptions about lifelong protection when even temporary protection is almost completely unproven. While health authorities assert that booster doses are "not currently recommended" for immunocompetent individuals, this recommendation is based on incomplete data and theoretical immunological memory rather than rigorous long-term efficacy studies. The package insert's admission that "the duration of the protective effect of RECOMBIVAX HB in healthy vaccinees is unknown at present and the need for booster doses is not yet defined" should raise serious questions about universal infant vaccination policies. Natural infection with hepatitis B virus (if cleared) provides lifelong immunity, whereas vaccine-induced immunity appears to wane in many individuals, potentially creating a false sense of security and unknown lifetime risk. The recommendation to vaccinate all infants at birth—including those born to HBsAg-negative mothers in low-risk populations—is particularly questionable given the unknown duration of protection and the remote risk of hepatitis B exposure in early childhood for most infants in developed countries.
Carcinogenicity, Mutagenicity, Fertility Studies#
RECOMBIVAX HB has NOT been evaluated for its carcinogenic or mutagenic potential, or its potential to impair fertility. This represents a significant gap in safety data for a product administered to infants at birth and recommended universally. No developmental toxicity studies have been conducted. For a vaccine that is administered to virtually all newborns in the United States and many other countries, the absence of these fundamental safety studies is remarkable.
Vitamins, Minerals & Metabolic Effects#
RECOMBIVAX HB, like other vaccines containing aluminum adjuvants and recombinant proteins, can affect nutrient status and metabolic processes through multiple mechanisms. While not commonly discussed in mainstream vaccine literature, the biological effects of vaccine components and immune system activation have documented impacts on nutrient metabolism:
Impact: Depletes glutathione, cysteine, and methionine stores. Impairs detoxification capacity. Infants and individuals with genetic variations in glutathione synthesis pathways (MTHFR polymorphisms, GST defects) are at higher risk. Reduced glutathione levels affect immune function, increase oxidative damage, and impair cellular metabolism. Multiple aluminum-containing vaccines administered simultaneously compound this depletion.
Impact: Magnesium is critical for over 300 enzymatic reactions, nerve function, muscle contraction, DNA/RNA synthesis, and ATP production. Depletion can cause neurological symptoms, irritability, sleep disturbances, muscle spasms, and cardiac arrhythmias. Aluminum-magnesium competition is particularly concerning given magnesium's role in neurological function and aluminum's neurotoxicity.
Impact: Vitamin C is rapidly depleted during immune challenges. Neonates and infants have limited vitamin C reserves and relatively low antioxidant capacity. Depletion impairs immune function (paradoxically), collagen synthesis, wound healing, iron absorption, and increases susceptibility to oxidative damage. Some research suggests vitamin C supplementation before and after vaccination may reduce adverse reactions.
Impact: Essential for immune function, DNA synthesis, cell division, growth, and development. Transient zinc depletion can impair thymic function and T-cell development in infants during critical developmental windows. Zinc deficiency affects wound healing, growth, appetite, taste, and increases susceptibility to infections—ironically, the opposite of the vaccine's intended effect.
Impact: B vitamins serve as cofactors for neurotransmitter synthesis, DNA methylation, homocysteine metabolism, and myelin formation. Depletion can affect neurological development, immune response quality, and detoxification capacity. Individuals with MTHFR genetic variants have impaired ability to process synthetic folic acid and require methylated forms of folate and B12, making them potentially more vulnerable to vaccine-induced B-vitamin depletion.
Impact: Critical for antioxidant defense, thyroid hormone metabolism, immune function, and DNA synthesis. Deficiency increases susceptibility to oxidative damage, impairs immune response (counterproductive for vaccination), and affects thyroid function. Selenium status may affect vaccine response and adverse reaction risk.
Impact: Vitamin A deficiency impairs both innate and adaptive immunity, including antibody responses to vaccines. Ironically, marginal vitamin A status may reduce vaccine efficacy while adequate status may reduce adverse reactions. Vitamin A is critical for maintaining mucosal immunity—the first-line defense against pathogens that vaccines bypass through injection.
Impact: Vitamin D deficiency associated with increased autoimmune disease risk, exaggerated inflammatory responses, and potentially increased vaccine adverse reaction risk. Given aluminum adjuvant's association with autoimmune conditions and the role of vitamin D in immune regulation, adequate vitamin D status may be particularly important around vaccination. Many infants are vitamin D deficient, especially in winter months.
IMPORTANT NOTE: These effects are based on established principles of immunology, toxicology, and nutritional biochemistry. Individual responses vary based on baseline nutritional status, genetic factors (particularly methylation and detoxification pathway polymorphisms), gut health, concurrent vaccine administration, and overall health status. The medical establishment rarely discusses vaccine-induced nutrient depletion, yet the biochemical mechanisms are well-documented in scientific literature outside the vaccine context.
CRITICAL DISCLAIMER: This information is for educational and research purposes only and represents a hypothesis based on known biochemical interactions. It is not medical advice. Parents and individuals considering RECOMBIVAX HB should request complete package inserts, ingredient lists, and discuss individual risk-benefit analysis with their healthcare provider. You should never feel pressured to make a vaccination decision before being fully informed. Remember that the package insert states "the duration of the protective effect of RECOMBIVAX HB in healthy vaccinees is unknown at present and the need for booster doses is not yet defined"—this fundamental uncertainty about long-term efficacy should factor into any vaccination decision.
Approval History, Surveillance & Critical Context#
Historical Significance#
RECOMBIVAX HB was approved by the FDA in 1986, making it the first recombinant vaccine approved for human use. This marked a paradigm shift from plasma-derived vaccines (which carried theoretical risks of blood-borne pathogens) to genetically engineered vaccines produced in yeast. The previous hepatitis B vaccine (Heptavax-B, approved 1981) was derived from plasma of chronically infected individuals—an approach that raised safety concerns and supply limitations. RECOMBIVAX HB's recombinant DNA technology was hailed as a major advance in vaccine production, enabling unlimited production capacity without reliance on infected human blood products.
Universal Infant Vaccination Policy#
In 1991, the CDC's Advisory Committee on Immunization Practices (ACIP) recommended universal hepatitis B vaccination for all infants in the United States, making it the first vaccine universally recommended at birth. This policy was controversial for several reasons:
- Hepatitis B is primarily transmitted through sexual contact, injection drug use, and perinatal transmission from infected mothers—modes of transmission irrelevant to most newborns in low-prevalence countries
- At the time, hepatitis B prevalence in the U.S. was low (<0.5% in the general population)
- The policy was justified by difficulty in reaching high-risk adult populations and the belief that universal infant vaccination would eventually eliminate hepatitis B
- Duration of protection was unknown then and remains unknown now, raising questions about whether infants vaccinated at birth will be protected during adolescence and adulthood when risk of exposure increases
Vaccine Injury Reporting & Surveillance#
RECOMBIVAX HB is covered under the National Childhood Vaccine Injury Compensation Program (VICP) established in 1986. Adverse events should be reported to:
- VAERS (Vaccine Adverse Event Reporting System): 1-800-822-7967 or www.vaers.hhs.gov - A passive surveillance system that captures only an estimated 1-10% of actual adverse events according to various studies. Both healthcare providers and patients/parents can report.
- Merck Sharp & Dohme Corp: 1-877-888-4231 - Direct manufacturer reporting
Important Note on Reporting: The VAERS system is passive and voluntary, meaning adverse events are significantly underreported. Healthcare providers may not recognize or report vaccine adverse events, and many parents are unaware the system exists. Studies have estimated that fewer than 10% of adverse events are reported to VAERS, meaning the true incidence of adverse reactions may be 10-100 times higher than official reports suggest. Lack of reporting also means many potential safety signals go undetected.
Storage & Handling#
- Store at 2-8°C (36-46°F) - refrigeration required
- Protect from light
- Do NOT freeze - freezing destroys potency
- Stable at temperatures 0° to 25°C (32° to 77°F) for 72 hours - for temporary excursions only, not for routine storage or shipping
Presentation & Color Coding#
RECOMBIVAX HB is available in color-coded formulations to prevent dosing errors:
- Pediatric/Adolescent (5 mcg): Yellow cap and stripe on vials, yellow plunger rod on prefilled syringes
- Adult (10 mcg): Green cap and stripe on vials, green plunger rod on prefilled syringes
- Dialysis (40 mcg): Blue cap and stripe on vials