Atorvastatin | Lipitor – HMG-CoA Reductase Inhibitor (Statin)
Drug Information: Atorvastatin | Lipitor – Statin HMG-CoA Reductase Inhibitor / Statin ATORVASTATIN Brand Name: LIPITOR® (Pfizer) Generic Name: Atorvastatin Calcium Class: HMG-CoA Reductase Inhibitor...
- ATORVASTATIN
- Mechanism of Action
- What the Mechanism Affects
- 1. Muscle Damage (Myopathy and Rhabdomyolysis)
- 2. Liver Dysfunction and Hepatotoxicity
- 3. Increased Diabetes Risk and Glycemic Control Impairment
- 4. Cognitive Impairment and Neurological Effects
- 5. Hemorrhagic Stroke Risk in Certain Populations
- 6. Coenzyme Q10 (CoQ10) Depletion
- 7. Peripheral Neuropathy
- 8. Tendon Disorders and Rupture
- 9. Endocrine Disruption
- 10. Interstitial Lung Disease
- 11. Drug-Drug Interactions (Extensive)
- Summary of Mechanism Effects
- Brand Names
- Prescribed For
- When Atorvastatin Must NOT Be Used
- Warnings and Precautions
- Adverse Reactions & Side Effects
- MOST COMMON ADVERSE REACTIONS (≥2% and Greater Than Placebo) - Placebo-Controlled Trials (N=8,755)
- ADDITIONAL ADVERSE REACTIONS REPORTED IN PLACEBO-CONTROLLED STUDIES
- SERIOUS AND/OR LIFE-THREATENING ADVERSE REACTIONS (POSTMARKETING REPORTS)
- LABORATORY ABNORMALITIES
- SPECIFIC CLINICAL TRIAL DATA
- PEDIATRIC ADVERSE REACTIONS (Ages 10-17 years, HeFH, 26-week controlled + 26-week open-label)
- Significant Drug Interactions
- Active & Inactive Ingredients by Tablet Strength
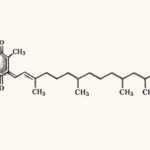
ATORVASTATIN#
Molecular Weight: 1209.42 (calcium salt)
Atorvastatin is an oral HMG-CoA reductase inhibitor (statin) approved by the FDA for treatment of hyperlipidemia and prevention of cardiovascular disease. The drug works by competitively inhibiting HMG-CoA reductase, the rate-limiting enzyme in cholesterol biosynthesis, thereby reducing cholesterol production in the liver and increasing hepatic LDL receptors to enhance LDL clearance. While marketed as effective for cardiovascular risk reduction and cholesterol management, atorvastatin carries significant risks including myopathy and rhabdomyolysis (potentially fatal muscle breakdown causing kidney failure), liver dysfunction with persistent transaminase elevations, increased risk of diabetes mellitus, cognitive impairment including memory loss and confusion, and hemorrhagic stroke in certain populations.
The drug is CONTRAINDICATED in pregnancy (Category X) and lactation due to potential fetal harm and passage into breast milk. Atorvastatin is extensively metabolized by CYP3A4, creating numerous clinically significant drug interactions that substantially increase myopathy risk. Patients taking cyclosporine, certain protease inhibitors, or other CYP3A4 inhibitors face exponentially higher risk of severe adverse effects. The 80 mg dose carries the highest risk profile and has been associated with increased rates of persistent liver enzyme elevations compared to lower doses. Critical monitoring of liver function tests and creatine phosphokinase (CPK) is essential, yet many adverse effects develop insidiously over months to years of use.
Mechanism of Action#
The Basic Mechanism#
Atorvastatin is a selective, competitive inhibitor of HMG-CoA reductase, the enzyme that catalyzes the conversion of HMG-CoA to mevalonate—an early and rate-limiting step in cholesterol biosynthesis. By blocking this enzyme in the liver, atorvastatin reduces intracellular cholesterol synthesis. In response to lowered intracellular cholesterol, the liver upregulates LDL receptors on hepatocyte surfaces, which increases the uptake and catabolism of circulating LDL particles from the bloodstream. This dual mechanism—reduced production and increased clearance—lowers plasma LDL-cholesterol levels.
In simple terms, it first slows down the liver’s own production of cholesterol by blocking a key step in how cholesterol is made. When the liver senses that its cholesterol level has dropped, it reacts by pulling more cholesterol out of the blood. It does this by putting more “catchers” (called LDL receptors) on its surface, which grab and remove LDL—often called bad cholesterol—from the bloodstream. So atorvastatin works both by making less cholesterol and by forcing the liver to pull it out of the blood, which leads to lower LDL (“bad”) cholesterol levels overall.
However, this artificial suppression of cholesterol synthesis has profound systemic effects beyond just lowering LDL-C numbers. Cholesterol is essential for:
- Cell membrane integrity and fluidity in every cell of the body
- Steroid hormone production including cortisol, aldosterone, testosterone, estrogen, and progesterone
- Bile acid synthesis for fat digestion and absorption
- Vitamin D synthesis in the skin
- Myelin sheath formation critical for nerve function
- Coenzyme Q10 (CoQ10) production essential for mitochondrial energy production
By inhibiting the mevalonate pathway, statins also block the synthesis of other biologically active substances derived from this pathway, including CoQ10, dolichol (required for protein glycosylation), heme A (for electron transport), and isoprenoids (for cell signaling). This explains many of the pleiotropic effects—both beneficial and harmful—observed with statin therapy.
The drug dosage, rather than systemic drug concentration, correlates better with LDL-C reduction because the primary site of action is the liver, where atorvastatin undergoes extensive first-pass metabolism. Approximately 70% of circulating HMG-CoA reductase inhibitory activity is attributed to active metabolites, particularly ortho- and para-hydroxylated derivatives.
Atorvastatin is extensively metabolized by cytochrome P450 3A4 (CYP3A4) in the liver and intestinal mucosa. This creates significant potential for drug-drug interactions with any medication or substance that inhibits or induces CYP3A4. The drug does NOT undergo significant renal elimination—less than 2% is recovered in urine—making it theoretically safer in renal impairment but creating dependence on hepatic function for clearance.
The absolute bioavailability of atorvastatin is approximately 14% due to extensive first-pass metabolism, but the systemic availability of HMG-CoA reductase inhibitory activity is approximately 30% due to active metabolites. The drug is highly protein-bound (≥98%) and has a mean elimination half-life of approximately 14 hours, though the half-life of HMG-CoA reductase inhibitory activity is 20-30 hours due to contributions from active metabolites.
What the Mechanism Affects#
1. Muscle Damage (Myopathy and Rhabdomyolysis)#
What This Means:
Muscle cells become energy-depleted and damaged. This manifests as myalgia (muscle pain), myopathy (muscle weakness with elevated CPK), or in severe cases, rhabdomyolysis—a life-threatening condition where muscle tissue breaks down rapidly, releasing myoglobin into the bloodstream that can cause acute kidney failure and death. Even “mild” muscle symptoms can significantly impair quality of life and physical function.
- Frequency: Myalgia occurs in 3.5-11% of patients in clinical trials, though real-world rates may be higher (10-25%). Severe myopathy with CPK >10x ULN occurs in ~0.1-0.2% of patients. Rhabdomyolysis is rare but has been reported with fatal outcomes.
- Risk factors: Advanced age (>65 years), female gender, small body frame, renal impairment, hepatic dysfunction, hypothyroidism, diabetes, excessive alcohol use, major surgery, trauma, severe infection, hypotension, and concomitant use of drugs affecting statin metabolism (especially CYP3A4 inhibitors).
- Dose-dependent: The 80 mg dose carries significantly higher myopathy risk than lower doses. In the TNT trial, more discontinuations occurred with 80 mg (9.9%) vs 10 mg (8.1%) due to adverse reactions.
- Immune-mediated necrotizing myopathy (IMNM): A rare autoimmune condition has been reported where muscle damage persists even after statin discontinuation, requiring immunosuppressive therapy. This suggests statins may trigger autoantibodies against muscle proteins.
- Subclinical effects: Many patients experience fatigue, reduced exercise tolerance, or subtle weakness without dramatic CPK elevations—effects that may not be recognized as statin-related but significantly impact daily functioning.
2. Liver Dysfunction and Hepatotoxicity#
What This Means:
Liver cells become damaged, leading to elevation of liver enzymes (transaminases) that leak from damaged hepatocytes into the bloodstream. In most cases this is asymptomatic and detected only on blood tests. However, progression to clinically significant hepatitis, cholestatic jaundice, or fulminant hepatic necrosis has been reported, with some fatal outcomes.
- Frequency: Persistent elevations in hepatic transaminases (>3x ULN on two or more occasions) occurred in 0.7% of patients in clinical trials, with rates of 0.2% (10 mg), 0.2% (20 mg), 0.6% (40 mg), and 2.3% (80 mg). The 80 mg dose carries >10-fold higher risk than 10 mg.
- Clinical presentation: Most cases are asymptomatic elevations detected on routine monitoring. Symptomatic hepatitis presents with fatigue, anorexia, right upper quadrant abdominal pain, dark urine, and jaundice.
- Fatal hepatic failure: Rare postmarketing reports of fatal and non-fatal hepatic failure have occurred. One patient in clinical trials developed jaundice.
- Alcohol interaction: Atorvastatin should be used with caution in patients who consume substantial quantities of alcohol and/or have a history of liver disease, as these factors increase hepatotoxicity risk.
- Monitoring: FDA recommends liver enzyme testing before initiating therapy and as clinically indicated thereafter. If serious liver injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs, atorvastatin must be promptly interrupted and not restarted.
3. Increased Diabetes Risk and Glycemic Control Impairment#
What This Means:
Statin therapy increases the risk of developing new-onset type 2 diabetes mellitus in patients without diabetes and worsens glycemic control in patients with existing diabetes. Increases in HbA1c and fasting serum glucose levels have been consistently reported with HMG-CoA reductase inhibitors including atorvastatin.
- Evidence from clinical trials: In SPARCL trial, diabetes was reported as an adverse reaction in 144 subjects (6.1%) in the atorvastatin 80 mg group vs 89 subjects (3.8%) in the placebo group—a 60% relative increase.
- Meta-analysis data: Large meta-analyses have shown statins increase new-onset diabetes risk by approximately 9-13%, with higher-intensity statin therapy (such as atorvastatin 80 mg) associated with greater risk than moderate-intensity therapy.
- Risk factors: Older age, obesity, metabolic syndrome, prediabetes (impaired fasting glucose or glucose tolerance), Asian ethnicity, female gender, and use of higher statin doses all increase diabetes risk.
- Clinical significance: The development of diabetes is a serious chronic condition requiring lifelong management and associated with increased risk of cardiovascular disease, kidney disease, neuropathy, retinopathy, and amputation.
- Mechanism uncertainty: The exact mechanisms remain incompletely understood, highlighting gaps in knowledge about statin effects on metabolic pathways beyond cholesterol.
4. Cognitive Impairment and Neurological Effects#
What This Means:
Patients may experience memory loss, forgetfulness, amnesia, memory impairment, confusion, and cognitive dysfunction. These effects have been reported across all statins and are generally reversible upon discontinuation, though the time to symptom onset varies from 1 day to years, and time to resolution ranges from days to weeks (median 3 weeks).
- FDA acknowledgment: The FDA has added warnings about cognitive impairment to statin labels based on postmarketing surveillance. Reports are generally non-serious and reversible.
- Clinical presentation: Patients report “brain fog,” difficulty concentrating, short-term memory problems, confusion, and slower cognitive processing. These symptoms can be frightening and significantly impair work performance and daily activities.
- Individual variability: Not all patients experience cognitive effects, and the severity varies widely. Some patients have profound impairment while others notice no changes.
- Diagnostic challenge: Cognitive symptoms are often attributed to aging, stress, or other conditions rather than recognized as statin-related, leading to underreporting and delayed discontinuation.
- Research limitations: Long-term cognitive outcomes of statin use remain incompletely studied, particularly regarding potential cumulative effects over decades of therapy.
5. Hemorrhagic Stroke Risk in Certain Populations#
What This Means:
In the SPARCL trial of patients with recent stroke or TIA (but no coronary heart disease), atorvastatin 80 mg was associated with a significantly higher incidence of hemorrhagic stroke compared to placebo. This represents an important harm that offsets some of the benefit in preventing ischemic stroke.
- SPARCL trial data: Hemorrhagic stroke occurred in 55 patients (2.3%) in the atorvastatin 80 mg group vs 33 patients (1.4%) in the placebo group (HR 1.68, 95% CI 1.09-2.59, p=0.0168). This represents a 68% relative increase in hemorrhagic stroke risk.
- Fatal vs non-fatal: The incidence of fatal hemorrhagic stroke was similar between groups (17 vs 18), but non-fatal hemorrhagic stroke was significantly higher with atorvastatin (38 patients, 1.6%) vs placebo (16 patients, 0.7%).
- Risk factors: Baseline characteristics including hemorrhagic stroke and lacunar stroke on study entry were associated with higher incidence of hemorrhagic stroke in the atorvastatin group.
- Population specificity: This increased risk was observed specifically in patients without CHD but with recent cerebrovascular events, raising questions about risk-benefit balance in this population.
- Dose relationship: The risk was observed with the 80 mg dose; relationship with lower doses in this population is unclear.
6. Coenzyme Q10 (CoQ10) Depletion#
What This Means:
Statin therapy reduces CoQ10 levels by 25-50% within weeks of starting treatment. This depletion impairs cellular energy production and antioxidant defenses, potentially contributing to multiple adverse effects including muscle symptoms, fatigue, heart failure, and cognitive dysfunction.
- Universal effect: All statins deplete CoQ10 because they all inhibit the mevalonate pathway. The degree of depletion is dose-dependent and correlates with the potency of cholesterol lowering.
- Clinical manifestations: Fatigue (often profound), muscle weakness and pain, exercise intolerance, shortness of breath, and possibly heart failure symptoms. Many patients report chronic tiredness that significantly impacts quality of life.
- Heart failure concern: The heart has extremely high energy demands and CoQ10 requirements. Some evidence suggests statins may worsen outcomes in patients with heart failure, possibly related to CoQ10 depletion. In TNT trial, hospitalization for heart failure showed a trend toward increased risk with 80 mg vs 10 mg dose.
- Supplementation debate: CoQ10 supplementation is not routinely recommended by conventional guidelines, though some evidence suggests it may help reduce muscle symptoms and fatigue. The FDA does not require CoQ10 depletion to be mentioned in statin prescribing information.
- Individual variability: Genetic variations in CoQ10 synthesis enzymes may make some individuals more susceptible to symptomatic CoQ10 depletion on statins.
7. Peripheral Neuropathy#
What This Means:
Patients may develop numbness, tingling, burning sensations, pain, or weakness in the extremities. Peripheral neuropathy has been reported with atorvastatin in postmarketing surveillance and can be debilitating, affecting mobility and quality of life.
- Postmarketing reports: Peripheral neuropathy is listed among adverse reactions identified during post-approval use of atorvastatin. The frequency is unknown but appears to be uncommon.
- Reversibility variable: Some cases resolve upon statin discontinuation, but others persist, suggesting potential for permanent nerve damage in susceptible individuals.
- Diagnostic challenge: Neuropathy symptoms develop gradually and may be attributed to other causes (diabetes, aging, vitamin deficiencies) rather than recognized as statin-induced.
- Distal symmetrical pattern: Typical presentation is symmetrical sensory symptoms starting in the feet and progressing proximally—the classic “stocking-glove” distribution.
8. Tendon Disorders and Rupture#
What This Means:
Tendon problems including tendinitis and tendon rupture have been reported with atorvastatin use. Tendon ruptures are serious injuries that often require surgical repair and prolonged rehabilitation, with potential for permanent functional impairment.
- Postmarketing data: Tendon rupture has been reported during post-approval use of atorvastatin. The frequency is unknown.
- Common sites: Achilles tendon is most commonly affected, but other tendons may be involved.
- Clinical presentation: Sudden pain, audible “pop,” and loss of function. May occur spontaneously or with minimal trauma.
- Risk factors: Advanced age, corticosteroid use, strenuous physical activity, rheumatoid arthritis, and renal impairment may increase risk.
9. Endocrine Disruption#
What This Means:
While clinical studies with atorvastatin have not shown it reduces basal plasma cortisol or impairs adrenal reserve in most patients, the theoretical concern remains. Caution should be exercised if atorvastatin is administered concomitantly with drugs that may decrease levels or activity of endogenous steroid hormones (such as ketoconazole, spironolactone, and cimetidine).
- Male fertility: The effects of statins on male fertility have not been studied in adequate numbers of patients. In animal studies at 100 mg/kg/day (16x human exposure at 80 mg dose), atorvastatin caused aplasia and aspermia in the epididymis, decreased sperm motility and spermatid head concentration, and increased abnormal sperm in male rats.
- Effects on women: The effects, if any, on the pituitary-gonadal axis in premenopausal women are unknown and have not been adequately studied.
- Testosterone concerns: Some observational studies have suggested statins may lower testosterone levels in men, though evidence is inconsistent. Symptoms of low testosterone include fatigue, decreased libido, erectile dysfunction, and reduced muscle mass—symptoms that overlap with other statin adverse effects.
- Clinical monitoring: Hormone levels are not routinely monitored in statin-treated patients, so endocrine effects may go undetected.
10. Interstitial Lung Disease#
What This Means:
Rare cases of interstitial lung disease have been reported with atorvastatin use, causing progressive shortness of breath, dry cough, and impaired oxygenation. This is a serious condition that can progress to pulmonary fibrosis if not recognized and the offending drug discontinued.
- Postmarketing reports: Interstitial lung disease is listed among adverse reactions identified during post-approval use of atorvastatin.
- Clinical presentation: Dyspnea (shortness of breath), non-productive cough, fatigue, and low-grade fever. Chest imaging shows interstitial infiltrates.
- Diagnosis: Requires high index of suspicion and often requires pulmonary consultation, high-resolution CT imaging, and sometimes lung biopsy.
- Management: Discontinuation of atorvastatin is essential. Corticosteroids may be required in severe cases.
11. Drug-Drug Interactions (Extensive)#
What This Means:
Numerous commonly prescribed medications significantly increase atorvastatin levels and toxicity risk. Some combinations are absolutely contraindicated, while others require dose limitations or extreme caution. The complexity of these interactions creates substantial risk for patients taking multiple medications.
- Cyclosporine: Increases atorvastatin AUC by 8.69-fold. Concomitant use should be AVOIDED.
- HIV protease inhibitors: Tipranavir + ritonavir increases atorvastatin AUC 9.36-fold (AVOID atorvastatin). Lopinavir + ritonavir, saquinavir + ritonavir, darunavir + ritonavir, fosamprenavir combinations: require lowest necessary dose or dose limitations. Nelfinavir: do not exceed 40 mg atorvastatin daily.
- HCV protease inhibitors: Glecaprevir + pibrentasvir increases atorvastatin AUC 8.28-fold (AVOID atorvastatin). Elbasvir + grazoprevir: do not exceed 20 mg atorvastatin daily. Simeprevir: use caution.
- Clarithromycin (antibiotic): Increases atorvastatin AUC 4.54-fold. Do not exceed 20 mg atorvastatin daily.
- Itraconazole (antifungal): Increases atorvastatin AUC 3.32-fold. Do not exceed 20 mg atorvastatin daily. Other azole antifungals likely have similar effects.
- Grapefruit juice: Contains components that inhibit CYP3A4. Increases atorvastatin AUC by 1.37-fold at 240 mL daily, but greater increases (up to 2.5-fold AUC) with excessive consumption (>1.2 liters/day). Patients should avoid grapefruit juice or limit to small occasional amounts.
- Gemfibrozil (fibrate): Concomitant use with atorvastatin should be AVOIDED due to increased myopathy/rhabdomyolysis risk. Other fibrates: use with caution and lowest dose necessary.
- Niacin (lipid-modifying doses ≥1 g/day): Increases risk of skeletal muscle effects. Use with caution.
- Colchicine: Cases of myopathy and rhabdomyolysis have been reported with atorvastatin co-administered with colchicine. Use with caution.
- Rifampin: Has dual effect—if given simultaneously with atorvastatin, AUC increases 1.12-fold, but if doses are separated, atorvastatin AUC decreases 80% (to 0.20). Simultaneous administration is recommended if combination needed.
- Oral contraceptives: Atorvastatin increases norethindrone AUC 1.28-fold and ethinyl estradiol AUC 1.19-fold. Consider when selecting contraceptive.
- Digoxin: Steady-state digoxin concentrations increased when co-administered with atorvastatin. Patients taking digoxin should be monitored appropriately.
- Warfarin: Atorvastatin had no clinically significant effect on prothrombin time, but patients should still be monitored when initiating or changing atorvastatin dose.
Summary of Mechanism Effects#
Atorvastatin’s mechanism of inhibiting HMG-CoA reductase creates a cascade of effects throughout the body far beyond simply lowering LDL cholesterol numbers. By blocking the mevalonate pathway, the drug interferes with production of cholesterol (essential for cell membranes, hormone synthesis, nerve function), CoQ10 (essential for cellular energy production), and other critical biomolecules. These wide-ranging metabolic disruptions explain the diverse adverse effect profile affecting muscles, liver, brain, nerves, tendons, and metabolic function.
The dose-dependent nature of both benefits and harms is clear, with the 80 mg dose showing substantially higher rates of adverse effects (persistent transaminase elevations: 2.3% vs 0.2% for 10 mg; more serious adverse reactions and discontinuations). The extensive CYP3A4-mediated metabolism creates numerous clinically significant drug interactions that dramatically increase toxicity risk, adding complexity to safe prescribing.
Bottom Line: While atorvastatin effectively lowers LDL cholesterol and has demonstrated reduction in cardiovascular events in certain populations, this comes at the cost of multiple systemic metabolic disruptions affecting energy production, hormone synthesis, muscle function, liver function, cognitive function, and more. The drug addresses a risk factor (high cholesterol) but does not address the underlying causes of cardiovascular disease (inflammation, oxidative stress, insulin resistance, endothelial dysfunction). An alternative approach would address root causes through comprehensive lifestyle modification (diet, exercise, stress management, sleep optimization) potentially combined with targeted nutritional support, rather than chronic pharmaceutical suppression of an essential biosynthetic pathway.
Brand Names#
Prescribed For#
- Prevention of Cardiovascular Disease in Adults Without Clinically Evident CHD: In adult patients without clinically evident coronary heart disease but with multiple risk factors for CHD (age, smoking, hypertension, low HDL-C, family history of early CHD), atorvastatin is indicated to reduce risk of: myocardial infarction, stroke, revascularization procedures, and angina.
- Prevention of Cardiovascular Disease in Adults With Type 2 Diabetes: In adult patients with type 2 diabetes without clinically evident CHD but with multiple risk factors (retinopathy, albuminuria, smoking, hypertension), atorvastatin is indicated to reduce risk of: myocardial infarction and stroke.
- Prevention in Adults With Clinically Evident CHD: In adult patients with clinically evident coronary heart disease, atorvastatin is indicated to reduce risk of: non-fatal myocardial infarction, fatal and non-fatal stroke, revascularization procedures, hospitalization for congestive heart failure, and angina.
- Primary Hyperlipidemia and Mixed Dyslipidemia (Adults): As adjunct to diet to reduce elevated total-C, LDL-C, apo B, and triglyceride levels and to increase HDL-C in adult patients with primary hypercholesterolemia (heterozygous familial and nonfamilial) and mixed dyslipidemia (Fredrickson Types IIa and IIb). Dose range: 10-80 mg once daily. Usual maintenance range: 10-20 mg daily. Patients requiring large LDL-C reduction (>45%) may be started at 40 mg daily.
- Hypertriglyceridemia (Fredrickson Type IV): As adjunct to diet for treatment of adult patients with elevated serum triglyceride levels.
- Primary Dysbetalipoproteinemia (Fredrickson Type III): For treatment of adult patients with primary dysbetalipoproteinemia who do not respond adequately to diet.
- Homozygous Familial Hypercholesterolemia (HoFH): To reduce total-C and LDL-C in patients with HoFH as adjunct to other lipid-lowering treatments (e.g., LDL apheresis) or if such treatments are unavailable. Dosage: 10-80 mg daily.
- Heterozygous Familial Hypercholesterolemia in Pediatric Patients: As adjunct to diet to reduce total-C, LDL-C, and apo B levels in pediatric patients 10-17 years of age with HeFH after failing adequate trial of diet therapy when: (a) LDL-C remains ≥190 mg/dL, OR (b) LDL-C remains ≥160 mg/dL AND there is positive family history of premature CVD OR two or more other CVD risk factors are present. Starting dose: 10 mg once daily. Usual dose range: 10-20 mg/day. Adjust at intervals of 4 weeks or more. Safety and effectiveness NOT established in pediatric patients under 10 years of age.
Limitations of Use: Atorvastatin has not been studied in conditions where the major lipoprotein abnormality is elevation of chylomicrons (Fredrickson Types I and V dyslipidemias). Therapy with lipid-altering agents should be only one component of multiple risk factor intervention. Drug therapy is recommended as adjunct to diet when response to diet restricted in saturated fat and cholesterol and other nonpharmacologic measures alone has been inadequate.
When Atorvastatin Must NOT Be Used#
ABSOLUTE CONTRAINDICATION: Active liver disease, which may include unexplained persistent elevations in hepatic transaminase levels. Atorvastatin can cause fulminant hepatic necrosis progressing to liver failure and death. Patients with active liver disease or unexplained persistent transaminase elevations must not receive atorvastatin.
ABSOLUTE CONTRAINDICATION: Atorvastatin is contraindicated in women who are pregnant or may become pregnant. Safety in pregnant women has not been established and there is no apparent benefit of lipid-lowering drugs during pregnancy. HMG-CoA reductase inhibitors decrease cholesterol synthesis and possibly synthesis of other biologically active substances derived from cholesterol, and may cause fetal harm when administered to pregnant women. Atorvastatin should be administered to women of childbearing potential only when they are highly unlikely to conceive and have been informed of potential hazards. If patient becomes pregnant while taking atorvastatin, the drug should be discontinued immediately.
ABSOLUTE CONTRAINDICATION: Atorvastatin use is contraindicated during breastfeeding. It is not known whether atorvastatin is present in human milk, but another drug in this class passes into human milk. Atorvastatin is present in rat milk. Because of potential for serious adverse reactions in breastfed infants, women must not breastfeed during treatment with atorvastatin.
ABSOLUTE CONTRAINDICATION: Known hypersensitivity to atorvastatin calcium or any component of the medication. Use in hypersensitive patients can precipitate anaphylaxis, angioedema, or severe anaphylactoid reactions.
Pregnancy Category X – Fetal Harm: Limited published data on atorvastatin use from observational studies, meta-analyses and case reports have not shown an increased risk of major congenital malformations or miscarriage. However, rare reports of congenital anomalies have been received following intrauterine exposure to other HMG-CoA reductase inhibitors. In animal reproduction studies, atorvastatin was not teratogenic in rats at doses up to 300 mg/kg/day or rabbits at doses up to 100 mg/kg/day (multiples of about 30x and 20x human exposure at MRHD of 80 mg based on surface area). However, in rats administered 225 mg/kg/day from gestation through lactation (22x human exposure), there was decreased pup survival at birth, postnatal day 4, weaning, and post-weaning; decreased pup body weight through postnatal day 91; and delayed pup development.
Females of Reproductive Potential – Contraception: Atorvastatin may cause fetal harm when administered to pregnant women. Advise females of reproductive potential to use effective contraception during treatment with atorvastatin and inform their healthcare provider of known or suspected pregnancy.
Warnings and Precautions#
⚠ SKELETAL MUSCLE EFFECTS (MYOPATHY AND RHABDOMYOLYSIS)#
Rare cases of rhabdomyolysis with acute renal failure secondary to myoglobinuria have been reported with atorvastatin and with other drugs in this class. A history of renal impairment may be a risk factor for development of rhabdomyolysis. Such patients merit closer monitoring for skeletal muscle effects.
Atorvastatin, like other statins, occasionally causes myopathy—defined as muscle aches or muscle weakness in conjunction with increases in creatine phosphokinase (CPK) values >10 times upper limit of normal (ULN). The concomitant use of higher doses of atorvastatin with certain drugs such as cyclosporine and strong cytochrome P450 3A4 (CYP3A4) inhibitors (e.g., clarithromycin, itraconazole, HIV and HCV protease inhibitors) increases the risk of myopathy/rhabdomyolysis.
Immune-Mediated Necrotizing Myopathy (IMNM): Rare reports of an autoimmune myopathy associated with statin use have been reported. IMNM is characterized by: proximal muscle weakness and elevated serum creatine kinase which persist despite discontinuation of statin treatment; muscle biopsy showing necrotizing myopathy without significant inflammation; improvement with immunosuppressive agents. This represents a serious autoimmune condition triggered by statin exposure that requires ongoing immunosuppressive therapy.
Clinical Recommendations: Myopathy should be considered in any patient with diffuse myalgias, muscle tenderness or weakness, and/or marked elevation of CPK. Patients should be advised to report promptly unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever or if muscle signs and symptoms persist after discontinuing atorvastatin. Atorvastatin therapy should be discontinued if markedly elevated CPK levels occur or myopathy is diagnosed or suspected.
Predisposing Factors for Myopathy/Rhabdomyolysis: Advanced age (>65 years), uncontrolled hypothyroidism, renal impairment, and concomitant use of certain interacting drugs (see Drug Interactions table). Lower starting and maintenance doses should be considered when atorvastatin is used concomitantly with drugs that increase atorvastatin levels.
Temporary Discontinuation: Atorvastatin therapy should be temporarily withheld or discontinued in any patient with an acute, serious condition suggestive of myopathy or having a risk factor predisposing to development of renal failure secondary to rhabdomyolysis (e.g., severe acute infection, hypotension, major surgery, trauma, severe metabolic/endocrine/electrolyte disorders, uncontrolled seizures).
Drug Interactions With Increased Myopathy/Rhabdomyolysis Risk:
- AVOID atorvastatin: Cyclosporine, tipranavir + ritonavir, glecaprevir + pibrentasvir
- Do not exceed 20 mg atorvastatin daily: Clarithromycin, itraconazole, saquinavir + ritonavir, darunavir + ritonavir, fosamprenavir, fosamprenavir + ritonavir, elbasvir + grazoprevir
- Do not exceed 40 mg atorvastatin daily: Nelfinavir
- Use with caution and lowest dose necessary: Lopinavir + ritonavir, simeprevir, fibric acid derivatives (especially gemfibrozil—avoid combination), erythromycin, azole antifungals, lipid-modifying doses of niacin (≥1 g/day), colchicine
⚠ LIVER DYSFUNCTION#
Statins, like some other lipid-lowering therapies, have been associated with biochemical abnormalities of liver function. Persistent elevations (>3 times ULN occurring on 2 or more occasions) in serum transaminases occurred in 0.7% of patients who received atorvastatin in clinical trials. The incidence of these abnormalities was dose-dependent: 0.2% (10 mg), 0.2% (20 mg), 0.6% (40 mg), and 2.3% (80 mg)—more than 10-fold higher rate with 80 mg compared to 10-20 mg doses.
One patient in clinical trials developed jaundice. Increases in liver function tests (LFT) in other patients were not associated with jaundice or other clinical signs or symptoms. Upon dose reduction, drug interruption, or discontinuation, transaminase levels returned to or near pretreatment levels without sequelae. Eighteen of 30 patients with persistent LFT elevations continued treatment with reduced dose of atorvastatin.
Hepatic Failure: There have been rare postmarketing reports of fatal and non-fatal hepatic failure in patients taking statins, including atorvastatin. If serious liver injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs during treatment with atorvastatin, promptly interrupt therapy. If an alternate etiology is not found, do not restart atorvastatin.
Monitoring Recommendations: It is recommended that liver enzyme tests be obtained prior to initiating therapy with atorvastatin and repeated as clinically indicated. Patients should be advised to report promptly any symptoms that may indicate liver injury including fatigue, anorexia, right upper abdominal discomfort, dark urine, or jaundice.
Alcohol: Atorvastatin should be used with caution in patients who consume substantial quantities of alcohol and/or have a history of liver disease.
⚠ USE IN PATIENTS WITH RECENT STROKE OR TIA#
In a post-hoc analysis of the Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) study where atorvastatin 80 mg vs placebo was administered in 4,731 subjects without CHD who had a stroke or TIA within the preceding 6 months, a higher incidence of hemorrhagic stroke was seen in the atorvastatin 80 mg group compared to placebo (55 events [2.3%] atorvastatin vs 33 events [1.4%] placebo; HR 1.68, 95% CI 1.09-2.59, p=0.0168).
The incidence of fatal hemorrhagic stroke was similar across treatment groups (17 vs 18 for atorvastatin and placebo groups, respectively). The incidence of non-fatal hemorrhagic stroke was significantly higher in the atorvastatin group (38 patients, 1.6%) compared to placebo group (16 patients, 0.7%). Some baseline characteristics, including hemorrhagic and lacunar stroke on study entry, were associated with a higher incidence of hemorrhagic stroke in the atorvastatin group.
⚠ ENDOCRINE FUNCTION#
Increases in HbA1c and fasting serum glucose levels have been reported with HMG-CoA reductase inhibitors, including atorvastatin. Statins interfere with cholesterol synthesis and theoretically might blunt adrenal and/or gonadal steroid production.
Clinical studies have shown that atorvastatin does not reduce basal plasma cortisol concentration or impair adrenal reserve. The effects of statins on male fertility have not been studied in adequate numbers of patients. The effects, if any, on the pituitary-gonadal axis in premenopausal women are unknown.
Caution should be exercised if a statin is administered concomitantly with drugs that may decrease the levels or activity of endogenous steroid hormones, such as ketoconazole, spironolactone, and cimetidine.
⚠ CNS TOXICITY (ANIMAL DATA)#
Brain hemorrhage was seen in a female dog treated for 3 months at 120 mg/kg/day (16x human exposure). Brain hemorrhage and optic nerve vacuolation were seen in another female dog sacrificed in moribund condition after 11 weeks of escalating doses up to 280 mg/kg/day. A single tonic convulsion was seen in each of 2 male dogs in a 2-year study.
CNS vascular lesions characterized by perivascular hemorrhages, edema, and mononuclear cell infiltration of perivascular spaces have been observed in dogs treated with other members of this class. A chemically similar drug in this class produced optic nerve degeneration (Wallerian degeneration of retinogeniculate fibers) in clinically normal dogs in dose-dependent fashion at doses producing plasma drug levels about 30 times higher than mean drug level in humans taking highest recommended dose.
Adverse Reactions & Side Effects#
MOST COMMON ADVERSE REACTIONS (≥2% and Greater Than Placebo) – Placebo-Controlled Trials (N=8,755)#
- Nasopharyngitis – 8.3% (any dose atorvastatin) vs 8.2% placebo; 12.9% (10 mg), 5.3% (20 mg), 7.0% (40 mg), 4.2% (80 mg)
- Arthralgia (joint pain) – 6.9% vs 6.5% placebo; dose-dependent: 8.9% (10 mg), 11.7% (20 mg), 10.6% (40 mg), 4.3% (80 mg)
- Diarrhea – 6.8% vs 6.3% placebo; notably higher with 40 mg dose: 7.3% (10 mg), 6.4% (20 mg), 14.1% (40 mg), 5.2% (80 mg)
- Pain in extremity – 6.0% vs 5.9% placebo; highest with 10 mg: 8.5% (10 mg), 3.7% (20 mg), 9.3% (40 mg), 3.1% (80 mg)
- Urinary tract infection – 5.7% vs 5.6% placebo; 6.9% (10 mg), 6.4% (20 mg), 8.0% (40 mg), 4.1% (80 mg)
- Dyspepsia – 4.7% vs 4.3% placebo; 5.9% (10 mg), 3.2% (20 mg), 6.0% (40 mg), 3.3% (80 mg)
- Nausea – 4.0% vs 3.5% placebo; higher with 40 mg: 3.7% (10 mg), 3.7% (20 mg), 7.1% (40 mg), 3.8% (80 mg)
- Musculoskeletal pain – 3.8% vs 3.6% placebo; 5.2% (10 mg), 3.2% (20 mg), 5.1% (40 mg), 2.3% (80 mg)
- Muscle spasms – 3.6% vs 3.0% placebo; 4.6% (10 mg), 4.8% (20 mg), 5.1% (40 mg), 2.4% (80 mg)
- Myalgia – 3.5% vs 3.1% placebo; highest with 40 mg: 3.6% (10 mg), 5.9% (20 mg), 8.4% (40 mg), 2.7% (80 mg)
- Insomnia – 3.0% vs 2.9% placebo; highest with 40 mg: 2.8% (10 mg), 1.1% (20 mg), 5.3% (40 mg), 2.8% (80 mg)
- Pharyngolaryngeal pain – 2.3% vs 2.1% placebo; 3.9% (10 mg), 1.6% (20 mg), 2.8% (40 mg), 0.7% (80 mg)
ADDITIONAL ADVERSE REACTIONS REPORTED IN PLACEBO-CONTROLLED STUDIES#
- Body as a whole: Malaise, pyrexia (fever)
- Digestive system: Abdominal discomfort, eructation (belching), flatulence, hepatitis, cholestasis
- Musculoskeletal system: Musculoskeletal pain, muscle fatigue, neck pain, joint swelling
- Metabolic and nutritional: Transaminases increase, liver function test abnormal, blood alkaline phosphatase increase, creatine phosphokinase increase, hyperglycemia
- Nervous system: Nightmare
- Respiratory system: Epistaxis (nosebleed)
- Skin and appendages: Urticaria (hives)
- Special senses: Vision blurred, tinnitus (ringing in ears)
- Urogenital system: White blood cells urine positive
SERIOUS AND/OR LIFE-THREATENING ADVERSE REACTIONS (POSTMARKETING REPORTS)#
- Anaphylaxis and angioneurotic edema – Severe allergic reactions including swelling that can obstruct airways; can be fatal
- Bullous rashes – Including erythema multiforme, Stevens-Johnson syndrome (potentially fatal), and toxic epidermal necrolysis (often fatal severe skin reactions)
- Rhabdomyolysis – Severe muscle breakdown causing kidney failure; can be fatal
- Myositis – Muscle inflammation
- Immune-mediated necrotizing myopathy (IMNM) – Autoimmune muscle disease persisting after statin discontinuation; requires immunosuppressive therapy
- Fatigue – Reported as adverse reaction in postmarketing surveillance; can be severe and debilitating
- Tendon rupture – Serious injury requiring surgical repair
- Fatal and non-fatal hepatic failure – Liver failure requiring transplant or resulting in death
- Dizziness, depression – Neuropsychiatric effects
- Peripheral neuropathy – Nerve damage causing numbness, tingling, pain, weakness in extremities
- Pancreatitis – Inflammation of pancreas; can be life-threatening
- Interstitial lung disease – Serious pulmonary condition
- Cognitive impairment – Memory loss, forgetfulness, amnesia, memory impairment, confusion. Reported for all statins. Reports generally non-serious and reversible upon statin discontinuation, with variable times to symptom onset (1 day to years) and symptom resolution (median 3 weeks).
LABORATORY ABNORMALITIES#
- Persistent transaminase elevations (≥3x ULN twice within 4-10 days): 1.3% with atorvastatin 80 mg vs 0.2% with atorvastatin 10 mg (TNT trial); 0.9% with atorvastatin 80 mg vs 0.1% with placebo (SPARCL trial)
- Elevations of CPK (≥10x ULN): Low overall but higher with high-dose atorvastatin (0.3% for 80 mg vs 0.1% for 10 mg in TNT trial; 0.1% for 80 mg vs 0.0% for placebo in SPARCL trial)
- Decreased hemoglobin/hematocrit: Small decreases (0.4 g% hemoglobin, 1.3 vol% hematocrit) in ~10% of patients; rarely clinically important
- Elevated liver enzymes/serum bilirubin: Rare; 0.3-1.0% range; warrants discontinuation
- Hyponatremia: Documented in postmarketing surveillance; potentially serious
SPECIFIC CLINICAL TRIAL DATA#
ASCOT Trial (10 mg daily, 3.3 years median follow-up)#
- Safety and tolerability profile comparable to placebo in 10,305 hypertensive participants
CARDS Trial (10 mg daily, 3.9 years median follow-up)#
- No difference in overall frequency of adverse or serious adverse reactions between atorvastatin and placebo in 2,838 subjects with type 2 diabetes
- No cases of rhabdomyolysis reported
TNT Trial (80 mg vs 10 mg daily, 4.9 years median follow-up)#
- More serious adverse reactions in high-dose group: 1.8% (80 mg) vs 1.4% (10 mg)
- More discontinuations due to adverse reactions in high-dose group: 9.9% (80 mg) vs 8.1% (10 mg)
- Persistent transaminase elevations (≥3x ULN twice): 1.3% (80 mg) vs 0.2% (10 mg)
- Elevations of CPK (≥10x ULN): 0.3% (80 mg) vs 0.1% (10 mg)
IDEAL Trial (80 mg daily, 4.8 years median follow-up)#
- No difference in overall frequency of adverse or serious adverse reactions between atorvastatin 80 mg and simvastatin 20-40 mg in 8,888 subjects with CHD
SPARCL Trial (80 mg daily, 4.9 years median follow-up)#
- Higher incidence of persistent hepatic transaminase elevations (≥3x ULN twice): 0.9% (atorvastatin) vs 0.1% (placebo)
- Elevations of CPK (>10x ULN): 0.1% (atorvastatin) vs 0.0% (placebo)
- Diabetes as adverse reaction: 6.1% (atorvastatin) vs 3.8% (placebo)
- Increased hemorrhagic stroke: 2.3% (atorvastatin) vs 1.4% (placebo) – see Warnings
- All-cause mortality: No significant difference between groups (9.1% atorvastatin vs 8.9% placebo)
- Cardiovascular death: Numerically smaller in atorvastatin group (3.3%) than placebo (4.1%)
- Non-cardiovascular death: Numerically larger in atorvastatin group (5.0%) than placebo (4.0%)
PEDIATRIC ADVERSE REACTIONS (Ages 10-17 years, HeFH, 26-week controlled + 26-week open-label)#
- Safety and tolerability profile generally similar to placebo in 187 boys and postmenarchal girls
- In limited controlled study, no significant effect on growth or sexual maturation in boys or on menstrual cycle length in girls
- Long-term efficacy NOT established: The long-term efficacy of atorvastatin therapy initiated in childhood to reduce morbidity and mortality in adulthood has not been established
⚠ CRITICAL SAFETY NOTE: The adverse reactions listed above represent documented occurrences in clinical trials and postmarketing surveillance. Serious and fatal outcomes have occurred including rhabdomyolysis with acute renal failure, fatal and non-fatal hepatic failure, hemorrhagic stroke, and severe hypersensitivity reactions. The high dose (80 mg) consistently demonstrates substantially higher rates of serious adverse effects compared to lower doses across multiple measures.
Many adverse effects develop insidiously over months to years of treatment and may not be immediately recognized as drug-related, including: cognitive impairment, peripheral neuropathy, chronic fatigue, exercise intolerance, new-onset diabetes, tendon problems, and subtle muscle weakness without dramatic CPK elevations. Real-world rates of muscle symptoms and other adverse effects may be substantially higher than clinical trial data suggest due to selection bias in trial enrollment, exclusion of high-risk patients, and limited duration of controlled trials relative to decades-long real-world use.
The extensive drug interaction profile creates substantial risk for patients taking multiple medications—a common scenario in the populations most likely to be prescribed statins (elderly, diabetic, cardiovascular disease patients). Concomitant use with CYP3A4 inhibitors or other interacting drugs can increase atorvastatin exposure many-fold, dramatically increasing adverse effect risk.
Significant Drug Interactions#
The risk of myopathy during treatment with statins is increased with concurrent administration of fibric acid derivatives, lipid-modifying doses of niacin, cyclosporine, or strong CYP3A4 inhibitors. Atorvastatin is metabolized by cytochrome P450 3A4 (CYP3A4). Concomitant administration of atorvastatin with strong inhibitors of CYP3A4 can lead to significant increases in plasma concentrations of atorvastatin. Atorvastatin is also a substrate of hepatic transporters OATP1B1, OATP1B3, and BCRP.
AVOID atorvastatin: Tipranavir + ritonavir (increases atorvastatin AUC 9.36-fold, Cmax 8.58-fold)
Do not exceed 20 mg atorvastatin daily: Saquinavir + ritonavir (increases AUC 3.93-fold, Cmax 4.31-fold – though actual clinical dose produces higher exposure than studied), darunavir + ritonavir (increases AUC 3.45-fold, Cmax 2.25-fold), fosamprenavir (increases AUC 2.53-fold, Cmax 2.84-fold), fosamprenavir + ritonavir (increases AUC 2.30-fold, Cmax 4.04-fold)
Do not exceed 40 mg atorvastatin daily: Nelfinavir (increases AUC 1.74-fold, Cmax 2.22-fold)
Use lowest dose necessary: Lopinavir + ritonavir
When co-prescribing atorvastatin with protease inhibitors, appropriate clinical assessment is recommended to ensure lowest dose necessary is used.
AVOID atorvastatin: Glecaprevir + pibrentasvir (increases atorvastatin AUC 8.28-fold, Cmax 22.00-fold due to BCRP, OATP1B1/1B3, and CYP3A inhibition). Concomitant use is NOT RECOMMENDED.
Do not exceed 20 mg atorvastatin daily: Elbasvir + grazoprevir (increases atorvastatin AUC 1.94-fold, Cmax 4.34-fold due to BCRP, OATP1B1/1B3, and CYP3A inhibition)
Use with caution: Simeprevir (increases AUC 2.12-fold, Cmax 1.70-fold), Telaprevir (increases AUC 7.88-fold, Cmax 10.60-fold), Boceprevir (increases AUC 2.32-fold, Cmax 2.66-fold)
⚠ Clinical Summary of Drug Interactions#
The extensive list of drug interactions with atorvastatin creates substantial complexity in safe prescribing, particularly for elderly patients and those with multiple comorbidities requiring polypharmacy. The most clinically significant interactions involve CYP3A4 inhibitors and drugs affecting hepatic transporters (OATP1B1, OATP1B3, BCRP), which can increase atorvastatin exposure by 2-fold to >10-fold, dramatically elevating myopathy and rhabdomyolysis risk.
Multiple commonly prescribed medications are either contraindicated with atorvastatin or require dose limitations: cyclosporine and certain protease inhibitors (tipranavir/ritonavir, glecaprevir/pibrentasvir) require complete avoidance; many HIV and HCV protease inhibitors, clarithromycin, and itraconazole require limiting atorvastatin to ≤20 mg daily; nelfinavir requires limiting to ≤40 mg daily. Yet patients are often prescribed these combinations without appropriate dose adjustments or adequate counseling about risks.
The combination of atorvastatin with gemfibrozil should be avoided entirely due to severe myopathy risk. Other fibrates, niacin (at lipid-lowering doses), and colchicine increase muscle toxicity risk and require caution with lowest necessary doses.
Patients should be explicitly counseled to inform all healthcare providers that they are taking atorvastatin before any new medication is prescribed, and to avoid grapefruit juice (or limit to occasional small amounts). The complexity of interactions, combined with the serious consequences of enhanced drug exposure (rhabdomyolysis with potential fatal acute renal failure), demands vigilant medication reconciliation and proactive patient education—yet these often receive inadequate attention in routine clinical practice.
Active & Inactive Ingredients by Tablet Strength#
ACTIVE INGREDIENT (All Strengths)#
Atorvastatin Calcium#
- 10 mg tablets: 10 mg atorvastatin per tablet (coded “PD 155” on one side and “10” on the other)
- 20 mg tablets: 20 mg atorvastatin per tablet (coded “PD 156” on one side and “20” on the other)
- 40 mg tablets: 40 mg atorvastatin per tablet (coded “PD 157” on one side and “40” on the other)
- 80 mg tablets: 80 mg atorvastatin per tablet (coded “PD 158” on one side and “80” on the other)
Chemical Description: Atorvastatin calcium is [R-(R*,R*)]-2-(4-fluorophenyl)-β,δ-dihydroxy-5-(1-methylethyl)-3-phenyl-4-[(phenylamino)carbonyl]-1H-pyrrole-1-heptanoic acid, calcium salt (2:1) trihydrate. The empirical formula is (C₃₃H₃₄FN₂O₅)₂Ca•3H₂O and its molecular weight is 1209.42. It is a white to off-white crystalline powder that is insoluble in aqueous solutions of pH 4 and below. Atorvastatin calcium is very slightly soluble in distilled water, pH 7.4 phosphate buffer, and acetonitrile; slightly soluble in ethanol; and freely soluble in methanol.
INACTIVE INGREDIENTS (ALL TABLET STRENGTHS)#
Common Inactive Ingredients (All Strengths)#
- Calcium carbonate, USP
- Candelilla wax, FCC
- Croscarmellose sodium, NF
- Hydroxypropyl cellulose, NF
- Lactose monohydrate, NF
- Magnesium stearate, NF
- Microcrystalline cellulose, NF
- Opadry White YS-1-7040 (hypromellose, polyethylene glycol, talc, titanium dioxide)
- Polysorbate 80, NF
- Simethicone emulsion
Tablet Appearance by Strength#
- 10 mg: White elliptical, film-coated tablets
- 20 mg: White elliptical, film-coated tablets
- 40 mg: White elliptical, film-coated tablets
- 80 mg: White elliptical, film-coated tablets
⚠ IMPORTANT INGREDIENT NOTES#
Lactose monohydrate: All tablet formulations contain lactose monohydrate as an inactive ingredient. Patients with rare hereditary problems of galactose intolerance, total lactase deficiency, or glucose-galactose malabsorption should not take atorvastatin tablets.
Magnesium stearate: Present in all formulations as a lubricant. Generally considered inert but may have minor effects on drug absorption or dissolution in sensitive individuals.
Polysorbate 80: A nonionic surfactant and emulsifier present in all formulations. Rarely, may cause hypersensitivity reactions in allergic individuals.
STORAGE & HANDLING#
- → Storage Temperature: Store at controlled room temperature 20-25°C (68-77°F) [see USP]
- → Protection Required: Protect from moisture and light
- → Container: Dispense in tight, light-resistant container