View Full Drug Profile: Loratadine (Including Mechanism of Action, and Side Effects)
Loratadine#
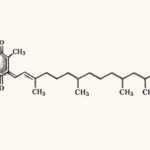
Loratadine is a widely used, long-acting second-generation antihistamine with selective peripheral H1-receptor antagonist activity. Developed as a successor to first-generation antihistamines, it is distinguished by its slightly decreased penetration of the blood-brain barrier, resulting in potent antihistaminic effects with less significant sedative or anticholinergic side effects common to older agents. It is prescribed for allergic conditions and is available over-the-counter in most countries.
Mechanism of Action#
What the Mechanism Affects#
Brand Names#
Prescribed For#
- Seasonal Allergic Rhinitis (Hay Fever): Relief of symptoms such as sneezing, rhinorrhea, itchy nose/palate/throat, and itchy/watery/red eyes.
- Perennial Allergic Rhinitis: Management of year-round allergic symptoms caused by indoor allergens like dust mites, pet dander, or mold.
- Chronic Idiopathic Urticaria: Treatment of red, itchy hives (wheals) of unknown cause, reducing pruritus and the number and size of hives.
- Other Allergic Skin Conditions: Used off-label to alleviate pruritus associated with mild allergic contact dermatitis or insect bite reactions.
- Adjunctive Therapy: Sometimes used alongside intranasal corticosteroids for more comprehensive allergic rhinitis control.
Contraindications & Black Box Warnings#
Absolute Contraindications & Major Precautions
Adverse Effects Profile#
Common Effects #
- Headache (most frequently reported, similar to placebo in some studies)
- Somnolence (Drowsiness) (less than 8%, significantly lower than 1st-gen antihistamines)
- Fatigue
- Dry Mouth
- Nervousness
- Gastrointestinal disturbances (e.g., nausea, gastritis)
Serious Side Effects#
- Cardiovascular: Tachycardia, palpitations, and QTc interval prolongation (rare, usually at high doses or with concurrent CYP inhibitors).
- Hypersensitivity: Severe reactions including anaphylaxis, angioedema, bronchospasm, urticaria, and rash.
- Hepatic: Elevated liver enzymes, hepatitis, jaundice (rare).
- Neurological: Seizures, dizziness, paresthesia, altered taste.
- Psychiatric: Insomnia, irritability, anxiety, depression (rare).
- Other: Alopecia, visual disturbances, menstrual disorders, breast enlargement (gynecomastia).
Drug Interactions#
Active & Inactive Ingredients#
The composition of a specific 10 mg tablet formulation of Loratadine is as follows[citation:2]:
Active Substance#
- Loratadine: 10 mg per tablet/reditab.
- Loratadine: 1 mg/mL in syrup.
Core Tablet Excipients (Inactive Ingredients)#
- Corn Starch
- Lactose Mnohydrate (dairy)
- Magnesium Stearate (a lubricant to aid manufacturing)
Syrup Excipients (Inactive Ingredients)#
- Citric Acid
- Edetate Disodium (Depletes calcium in the body) Made from combining ethylenediamine, formaldehyde, and sodium cyanide
- Artificial Flavor (not specified but likely methyl anthranilate)
- Glycerin (a binder and disintegrant)
- Propylene Glycol (solvent)
- Sodium Benzoate (preservative and toxin)
- Sugar
- Water
Reditabs Excipients (Inactive Ingredients)#
- Citric Acid
- Gelatin
- Mannitol (diuretic) Produced from high fructose corn syrup
- Mint flavor
Important Note: Different brands and formulations (e.g., syrup, orally disintegrating tablets) will have different inactive ingredient lists. Patients with specific allergies or intolerances (e.g., to lactose) should always check the full product information for their specific medication. This information is provided and intended to be accurate but always check with your doctor before starting or stopping any medications.
Clinical Trials#
Loratadine (marketed as CLARITIN) was approved by the FDA on the basis of clinical trials consisting of “either CLARITIN Tablets or another antihistamine and/or placebo in double-blind randomized controlled studies.” Notice that the placebo group could include an antihistamine. The placebo group also is not saline but rather contains toxins designed to create a similar AE profile, to insinuate safety.
Allergic Rhinitis Efficacy#
- Signature FDA Approval Study: In this study none of the participants reported a good to excellent response. And no difference was achieved in non-nasal symptoms. In otherwords, there was no real-world improvement in health.
Placebo Trials#
- None of the placebo trials contain saline. All of them contain toxins and some are even active drugs. For instance, several studies compare Loratadine with other drugs as placebos. This is intellectually dishonest, especially considering they hide these facts.
Source: Clinical trial data summarized from the official FDA label for CLARITIN (loratadine).
Vitamins & Minerals Depleted#
While not classically associated with severe vitamin deficiencies like some long-term medications, the mechanism of action and metabolic pathway of Loratadine does effect specific nutrients that patients should be aware of.
Mechanism: EDTA binds to di- and trivalent metal ions (e.g., Calcium, Zinc, Iron, Magnesium) in the gastrointestinal tract, forming soluble complexes that can reduce the absorption of these essential minerals.
Potential Impact: Reduced stomach acid can impair the breakdown and absorption of protein-bound nutrients, including Vitamin B12, Iron, and Calcium. Altered digestion may also affect the absorption of fats and fat-soluble vitamins (A, D, E, K).
Recommendations for Patients
- Monitor & Space Intake: If using the syrup long-term, consider spacing medication and meal/mineral supplement intake by 2-3 hours to minimize chelation.
- Consider Formulation: For chronic use, tablets or Reditabs may be preferable to syrup to avoid regular EDTA exposure.
- Diet Focus: Maintain a diet rich in whole foods, lean proteins, and colorful vegetables to support natural vitamin and mineral levels.
- Professional Guidance: Discuss long-term nutritional status with a healthcare provider, especially if symptoms like unusual fatigue, weakness, or bruising occur, which could indicate a deficiency.
Note: This information is based on pharmacological principles and ingredient analysis. Significant deficiency from Loratadine alone is uncommon but possible with prolonged use, poor diet, or pre-existing malabsorption conditions.
Loratadine (sold as Claritin, Allerclear) – Marketed under many different names including Allergy Relief and Alavert as well as Non-Drowsy Allergy. Side effects include liver damage, tightness in chest or difficulty breathing, fainting, and seizures as well as low platelet count which is not good.
A paper published in the Lancet looked at the dangers of non-sedating antihistamines. In this paper, the dangers are discussed and one of the things mentioned is the banning of terfenadine which is to all intents and purposes the same drug as Claritin. The reason it was banned is for life-threatening side effects. It also mentions the prevalence of arrhythmias among antihistamine users and how it is crucial not to exceed dose due to serious side effects.
[ref]Dangers of non-sedating antihistamines[/ref]
The drug manufacturer says they are rare but the research finds they are not so rare. What is most concerning to us is the discussion in regards to whether doctors should discuss risk with patients which could “unnecessarily alarm” them. You can’t trust a system that doesn’t care enough about you to give you the facts.